Introduction: Facial palsy affects both the functional and aesthetic aspects of the face. Many techniques for both static and dynamic facial reanimation have been described. Here, we present our series of facial reanimation using the functioning free gracilis muscle flap innervated by the masseteric nerve.
Materials and Methods: During the period of 2012-2017, facial reanimation in patients with facial palsy using the functioning free gracilis muscle flap innervated by the ipsilateral masseteric nerve was performed in 18 patients. The extent of oral commissure movement was determined by the measurements and comparison on both sides of the face from the auricular tragion to the oral commissure, both at rest and with a forced smile.
Results: All patients were followed up for at least 9 months and 17 out of 18 patients (94%) were either satisfied or very satisfied with their outcome. The measurements for oral commissure movement on the side of facial paralysis were found to be similar to that of the normal side in all the 18 cases.
Conclusion: Although it is not the first choice of facial reanimation in the current management of unilateral facial paralysis, since it doesn’t provide spontaneity, the masseteric nerve is a powerful and reliable facial reanimation donor nerve to innervate the functioning free gracilis muscle flap, which can restore the dynamic smile with minimal donor morbidity.
Facial paralysis results in an altered and reduced range of emotional expression of the face. The condition not only affects the appearance, but also the functional aspects of the face in terms of communication and oral competence. The management of facial paralysis involves a multi-disciplinary approach with contributions from various medical specialties and allied health, including physiotherapy and acupuncture.
The previous studies have shown that if the duration of injury to the nerve is less than two years, direct surgical intervention to the facial nerve, such as direct repair or nerve graft/transfer, is appropriate because the motor neural activity is still present [1,2]. In cases where injury to the facial nerve occurred over 2 years prior, the dynamic reanimation aims to restore a symmetrical and coordinated smile. Reanimation of the mid-face also increases tone in the cheek and facilitates speech and eating. In this study, we evaluate our use of the free gracilis muscle flap innervated by the masseter nerve for facial reanimation. Spira (1978) used the masseteric nerve to reinnervate the lower division of facial nerve [3]. In 1989, Zuker and Manteklow described using the motor branches of the trigeminal nerve as a direct motor donor source during free muscle transfer in the treatment of Moebius syndrome [4]. In 2004, Luis reported a successful case of masseteric nerve transfer in facial paralysis [5].
In the primary author’s home country (Vietnam), there have been reports of the two-stage approach for the treatment of facial palsy with free gracilis muscle transfer, but there are no published reports of facial reanimation based on the masseteric nerve. In this study, we evaluate the effectiveness of the masseter motor nerve in providing innervation to a gracilis functioning free muscle transplantation for facial palsy.
From the period of 2012 to 2017, 18 facial paralysis patients were treated with free gracilis muscle transplantation innervated by masseteric nerve and were included in the study. All the patients had facial paralysis present for more than 24 months. The causes of facial paralysis included post-surgical resection of acoustic neuroma and Bell’s palsy. Indications for various treatment methods for facial palsy within our institution were based principally on the time elapsed since the injury of the facial nerve (Table 1) [2].
Operative Technique
All the patients underwent surgery under general anesthesia and in a supine position. A free gracilis muscle flap was harvested based on its neurovascular bundle. The obturator nerve was harvested 10 cm in average. In general, 10-12 cm in length of the gracilis muscle was harvested and a subcutaneous pocket was made in the paralyzed side of the face from the zygoma to the lateral commissure of the lip. The masseteric nerve was identified in the deep layer of the masseter muscle. The gracilis flap was then inset proximally and distally into the pocket and its nerve coapted to the masseteric nerve (end-to-end), followed by arterial and venous anastomosis of the flap to their recipient vessels (Figure 1). Flap monitoring was assisted with hand-Doppler ultrasound and the patients stayed in the hospital for 5 to 7 days. Rehabilitation commenced one month after surgery. Postoperative follow-up was performed for at least 9 months after surgery.
Evaluation
The outcome of the study was collected through the patient outcome survey, clinical assessment (by the primary author), and the analysis of before-and-after surgery photographs and videos of patients at rest and with facial movement. The dynamic measurements were made on each side to evaluate the excursion of the oral commissure during a forced smile. We used the facial grading system proposed by Terzis in 1991 [6].
From 2012 to 2107, 18 facial palsy patients were treated using the functioning free gracilis muscle transplantation with the masseteric nerve serving as the donor innervation. There were 6 male and 12 female patients with a mean age of 23 years (range 8-54). The aetiology of facial paralysis is shown in Table 2.
The average operative time was 7 hours. All the gracilis flaps were viable. Reinnervation of the gracilis muscle was detected in all patients by 4 months, and 2 patients had signs of reinnervation earlier at 2.5 months. One case was complicated by postoperative haematoma on the first postoperative day. This was treated with suture removal and evacuation of the haematoma through the facial wound. There was no notable donor site morbidity. All the patients commenced surgeon-directed rehabilitation at 1 month after surgery. This included forced repetitive smiling in front of a mirror for at least 20 minutes per day.
The result of the patient survey evaluation of their outcome was good to excellent in most patients (Table 3). All the patients were able to move the commissure of the lip when they bit their teeth together. The mean oral commissural movement on the paralyzed side was 1.4 cm (range 1 to 1.7 cm).
Gracilis functioning free muscle transplantation has been used in facial palsy reanimation for over 30 years [6]. In the classic description, patients underwent a two-stage procedure, spaced at least 9-12 months apart. The current concept of the first choice neurotizer of unilateral paralysis is still the cross face nerve graft (CFNG), since it offers emotional spontaneity. However, the two-stage operations, higher complication rate, and the prolong rehabilitation time due to the relatively small number of myelinated axons make this procedure more complicated. The previous study showed that the axon density in distal CFNG was only 1/3 of masseter nerve [7]. Although the current studies focus on how to improve the functional result via either sensory (including end-to-end/end-to-side infraorbital nerve coaptation) or motor protection (dual innervation via CFNG and masseteric nerve), still there is no consensus [8,9].
The use of non-facial motor nerves as a donor source for reanimation in facial paralysis has been widely reported in the literature. The hypoglossal and spinal accessory nerves are two such examples of donor nerves used to restore and power the facial movement [10]. However, the use of the hypoglossal and spinal accessory nerves is disadvantaged by the sensory and atrophic changes to half the tongue and reduced strength to shoulder mobility, respectively. However, in some specific situation, such as bilateral Mobius syndrome, the bilateral spinal accessory nerve has been reported as the donor nerve [10].
The masseteric nerve is a branch of the mandibular division of the trigeminal nerve, which divides into two superior and inferior branches between the middle and deep layers of the masseter muscle [11]. In 2004, Kun Hwang and his colleagues examined the masseteric nerve on 48 cadavers that described the anatomical landmarks of the nerve [12]. Others have described the technical aspects to identify the masseteric nerve [6,8]. Coomb’s report [14] described the use of the masseteric nerve with no significant functional problems after surgery with chewing or biting. Nowadays, a one-stage masseteric nerve innervated free gracilis has thereby become the most popular using non-facial nerve as the neurotizer procedure for facial reanimation [15,16].
In the classic two-stage gracilis functioning free muscle transplantation with cross-facial nerve graft, as some authors have noted, the lip movement in the classically-performed procedure is often slow to regain, and the range of movement is frequently limited compared to the ipsilateral masseteric nerve as the donor. Moreover, the one-stage gracilis functioning free muscle transplantation with ipsilateral masseteric nerve also reduced surgical time [15,17,18]. In this study, the time after surgery to the commencement of oral commissure lip movement in the paralyzed side was rapid, beginning at 2.5 months on average. The degree of muscle movement achieved ranged from 10-17 mm. This compared favorably with the normal non-paralyzed side, where the commissure range of movement was measured at 7-22 mm (mean 14 mm) (Figure 2). The significant degree of movement achieved through using the masseteric nerve as the donor may be indicative of the high number of axons in this nerve (1543, almost three times that of the facial nerve) [14].
When using non-facial nerves (such as the masseteric) as donor sources for facial reanimation, it has been previously thought that the restored facial muscles are not able to move independently in relation with the associated muscle of the donor nerve. However, some reports show that the patients may be able to achieve the spontaneous smile due to the cortical adaptation [20]. In our series, some patients could move their lips autonomously, without any bite action, at 36 months postoperatively. It is thought that in young patients the spontaneity can be achieved easier.
Rehabilitation is vital to achieving a good outcome. After the first month after surgery, the patients in this study were instructed to perform exercises with a mirror for 20 minutes per day. The program included bite motion exercises as well as mental tasks associated with the bite. It is likely that during this period of recovery, the rehabilitation exercises encourage and activate connections between the facial nerve and trigeminal nerve centers [21]. The age of the patient at operation did not influence the result from an aesthetic or functional point of view.
Although the masseteric nerve is not the first choice of facial reanimation in the current management of unilateral facial paralysis, since it doesn’t provide spontaneity, it is a powerful and reliable facial reanimation donor nerve. The currently acceptable indications of using this nerve include (1) previous failure cases, (2) aged patients, and (3) bilateral palsy case such as Mobius syndrome [10,22]. Gracilis functioning free muscle flap innervated by the masseteric nerve can reliably restore dynamic smile movement and is associated with minimal donor morbidity.
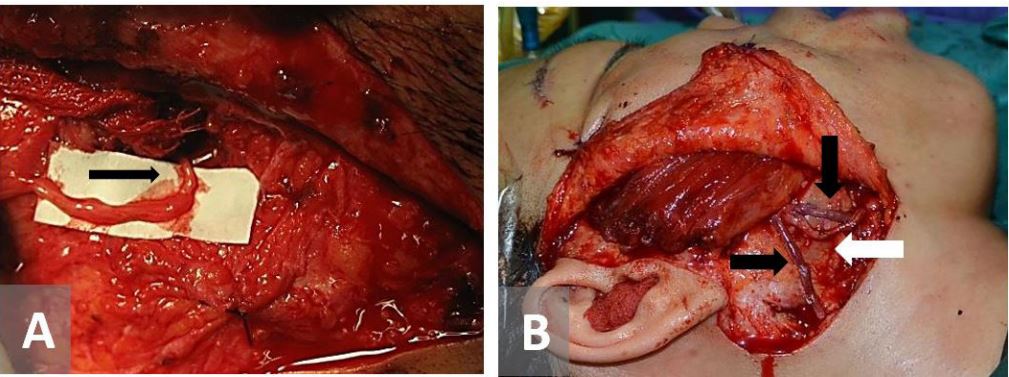
Figure 1. Free gracilis flap inset. (A) Masseteric-Obturator nerve coaptation. (B) Arterial (white arrow) and venous anastomosis (black arrows).
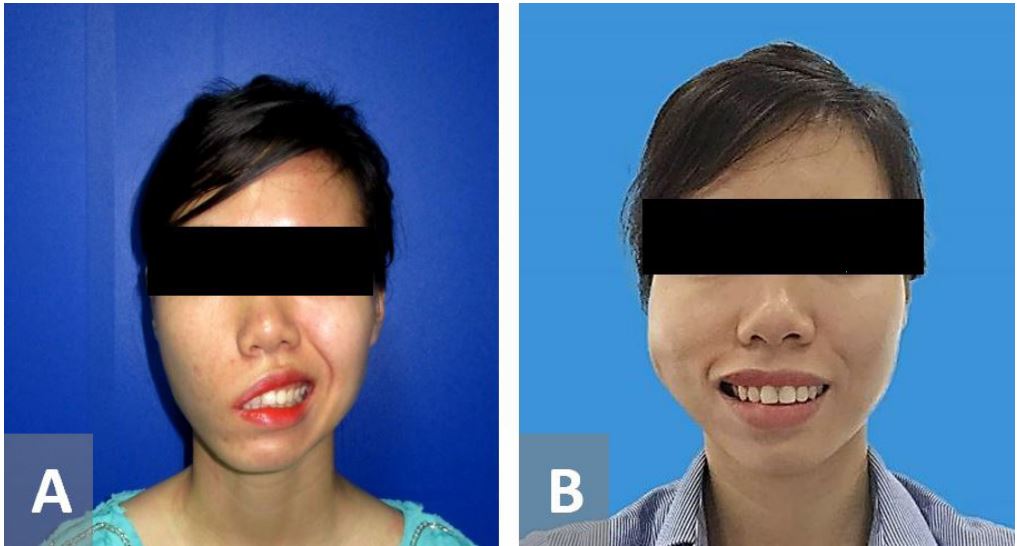
Figure 2. A 25-year-old female with Bell’s palsy on the right side for 22 years. The patient had a free gracilis flap innervated by the masseteric nerve. (A) Pre-operative figure. (B) 9 months after surgery with balance comissure movements on both sides.
Received date: June 30, 2018
Accepted date: April 08, 2019
Published date: July 11, 2019
© 2019 The Authors. This is an open-access article distributed under the terms of the Creative Commons Attribution 4.0 International License (CC-BY).
This case report describes authors successfully attempt to perform a topographically correct sural nerve transplantation in the extracranial facial nerve stem using the intraneural facial nerve stem topography proposed by Meissl in 1979. This is the first reported case of successful fascicular nerve grafting of the facial nerve stem following extensive laceration.
This article presents a groundbreaking surgical approach for treating facial paralysis, focusing on the combination of the pronator quadratus muscle (PQM) and the radial forearm flap (RFF). It addresses the challenges in restoring facial functions and skin closure in paralysis cases. The study's novelty lies in its detailed examination of the PQM's vascular anatomy when combined with the RFF, a topic previously unexplored. Through meticulous dissections, it provides crucial anatomical insights essential for enhancing facial reanimation surgeries, offering significant benefits in medical practices related to facial reconstruction and nerve transfer techniques.

Bui MA, Goh R, Vu TT. Dynamic reanimation of smile in facial paralysis with gracilis functioning free muscle flap innervated by masseteric nerve: The first Vietnamese series Int Microsurg J 2019;3(2):4. https://doi.org/10.24983/scitemed.imj.2019.00116